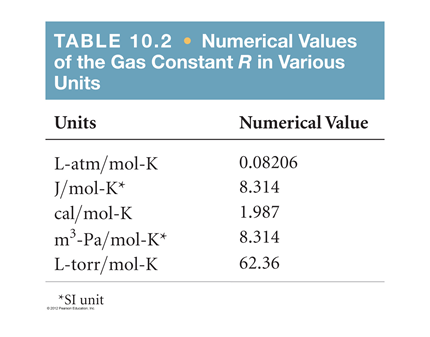
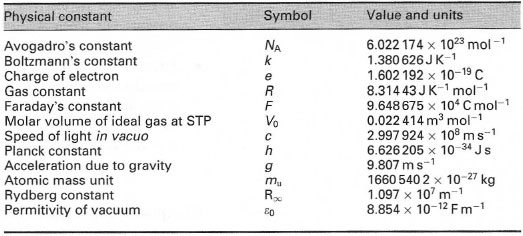
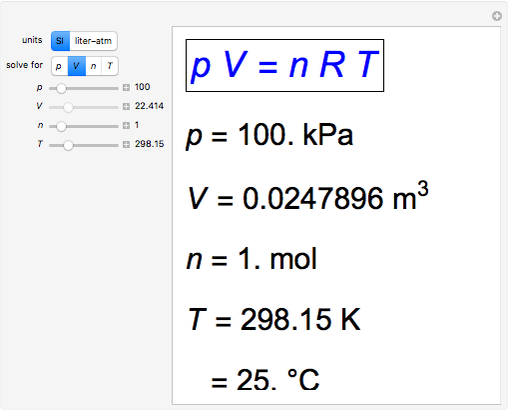
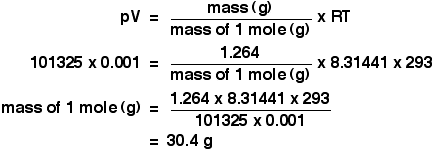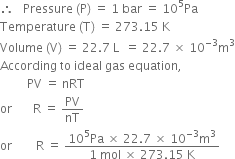Aaditional ideal gas - The equation of the state of an ideal gas as discussed PV = m (R) T The units - StuDocu

Chapter 5: Gases and the Kinetic - Molecular Theory 5.1 An Overview of the Physical States of Matter 5.2 Gas Pressure and it's Measurement 5.3 The Gas. - ppt download

SOLVED:Two moles an argon gas are energy of the gas_ temperature of 305 K. Calculate the average kinetic energy per atom Toot mean square (rms) speed atoms in the gas, and the

A Level Chemistry Measuring moles of GASES by measuring their volume, pressure and temperature PV = nRT. - ppt download