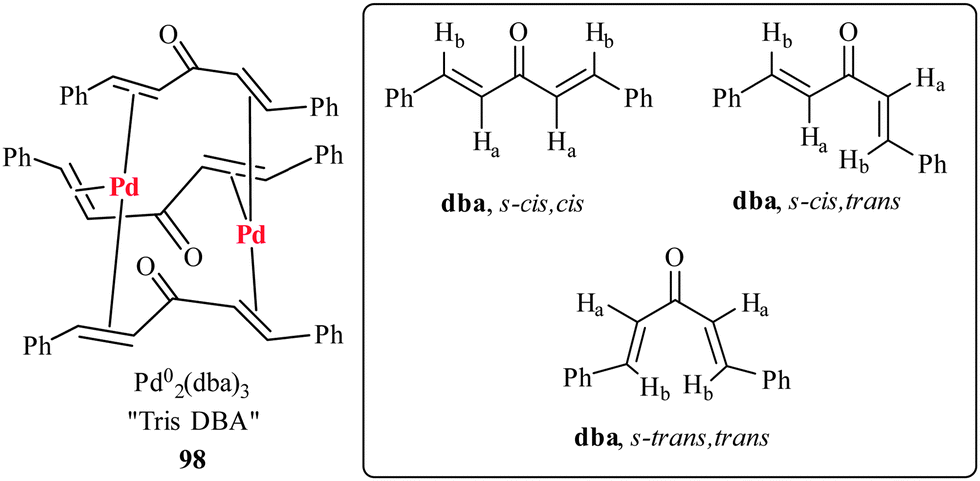
Exploiting Noninnocent (E,E)‐Dibenzylideneacetone (dba) Effects in Palladium(0)‐Mediated Cross‐Coupling Reactions: Modulation of the Electronic Properties of dba Affects Catalyst Activity and Stability in Ligand and Ligand‐Free Reaction Systems ...

Figure 15 from The elusive structure of Pd2(dba)3. Examination by isotopic labeling, NMR spectroscopy, and X-ray diffraction analysis: synthesis and characterization of Pd2(dba-Z)3 complexes. | Semantic Scholar

Exploiting Noninnocent (E,E)‐Dibenzylideneacetone (dba) Effects in Palladium(0)‐Mediated Cross‐Coupling Reactions: Modulation of the Electronic Properties of dba Affects Catalyst Activity and Stability in Ligand and Ligand‐Free Reaction Systems ...

Exploiting Noninnocent (E,E)‐Dibenzylideneacetone (dba) Effects in Palladium(0)‐Mediated Cross‐Coupling Reactions: Modulation of the Electronic Properties of dba Affects Catalyst Activity and Stability in Ligand and Ligand‐Free Reaction Systems ...
Anti-cancer palladium complexes: a focus on PdX 2 L 2 , palladacycles and related complexes - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C4CS00063C

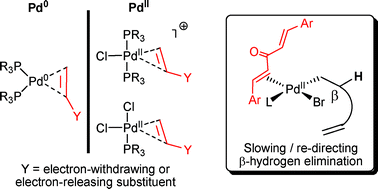
Palladium‐Catalysed Cross‐Coupling and Related Processes: Some Interesting Observations That Have Been Exploited in Synthetic Chemistry - McGlacken - 2009 - European Journal of Organic Chemistry - Wiley Online Library

The elusive structure of Pd2(dba)3. Examination by isotopic labeling, NMR spectroscopy, and X-ray diffraction analysis: synthesis and characterization of Pd2(dba-Z)3 complexes. | Semantic Scholar

Figure 10 from The elusive structure of Pd2(dba)3. Examination by isotopic labeling, NMR spectroscopy, and X-ray diffraction analysis: synthesis and characterization of Pd2(dba-Z)3 complexes. | Semantic Scholar

Exploiting Noninnocent (E,E)‐Dibenzylideneacetone (dba) Effects in Palladium(0)‐Mediated Cross‐Coupling Reactions: Modulation of the Electronic Properties of dba Affects Catalyst Activity and Stability in Ligand and Ligand‐Free Reaction Systems ...
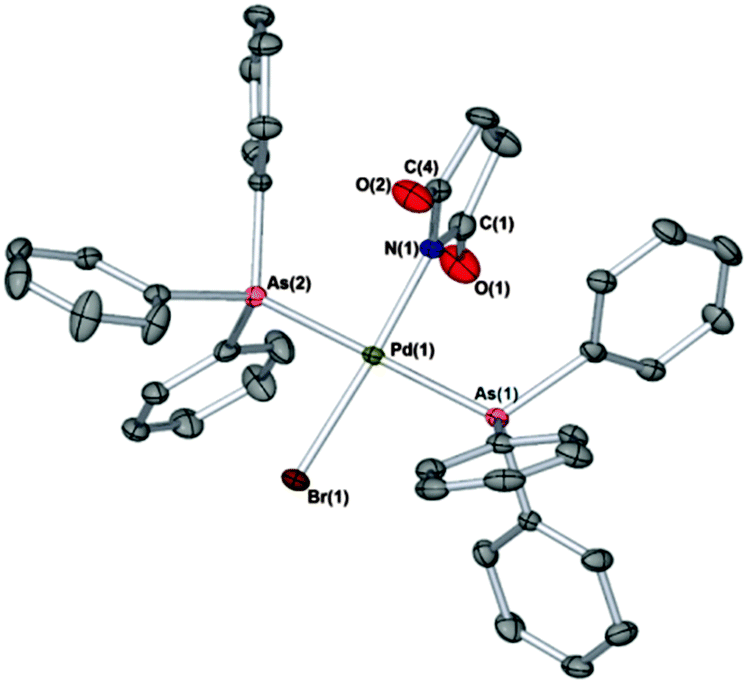
AsCat and FurCat: new Pd catalysts for selective room-temperature Stille cross-couplings of benzyl chlorides with organostannanes - Chemical Communications (RSC Publishing) DOI:10.1039/C4CC09810B

Figure 9 from The elusive structure of Pd2(dba)3. Examination by isotopic labeling, NMR spectroscopy, and X-ray diffraction analysis: synthesis and characterization of Pd2(dba-Z)3 complexes. | Semantic Scholar

Halide and pseudohalide effects in Pd-catalysed cross-coupling reactions - New Journal of Chemistry (RSC Publishing) DOI:10.1039/B605699G

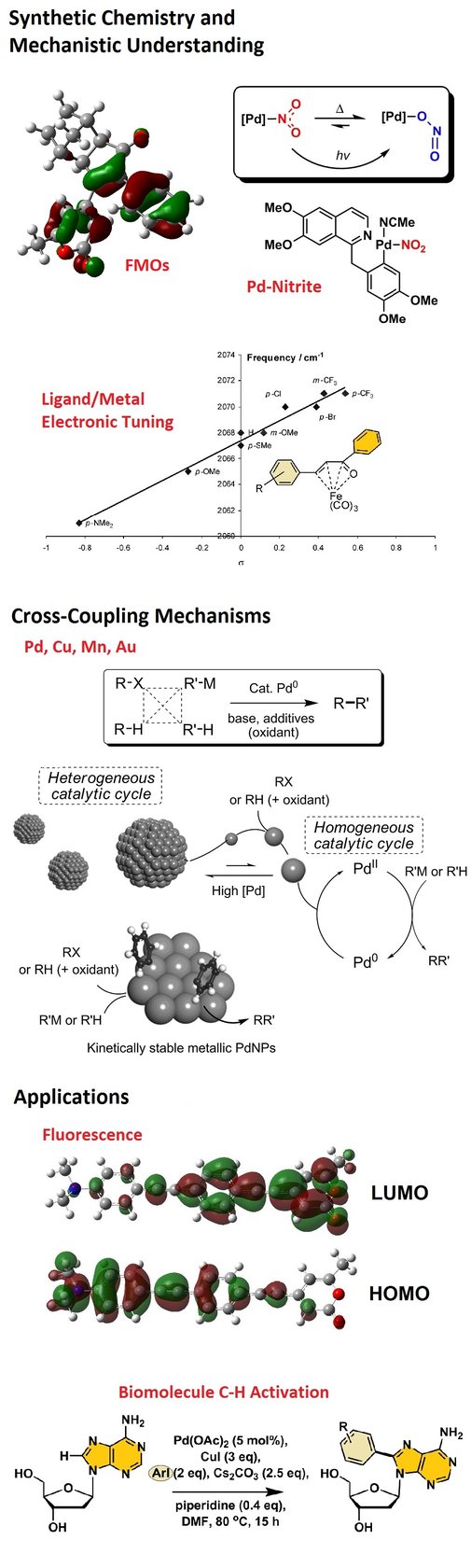
Redox‐Active NOx Ligands in Palladium‐Mediated Processes - Fairlamb - 2015 - Angewandte Chemie International Edition - Wiley Online Library

Palladium‐Catalysed Cross‐Coupling and Related Processes: Some Interesting Observations That Have Been Exploited in Synthetic Chemistry - McGlacken - 2009 - European Journal of Organic Chemistry - Wiley Online Library

Analysis of Stereochemical Convergence in Asymmetric Pd‐Catalysed Allylic Alkylation Reactions Complicated by Halide and Memory Effects - Fairlamb - 2002 - Chemistry – A European Journal - Wiley Online Library
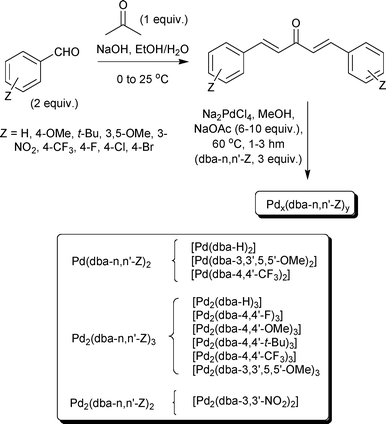
π-Acidic alkene ligand effects in Pd-catalysed cross-coupling processes: exploiting the interaction of dibenzylidene acetone (dba) and related ligand ... - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/B811772A