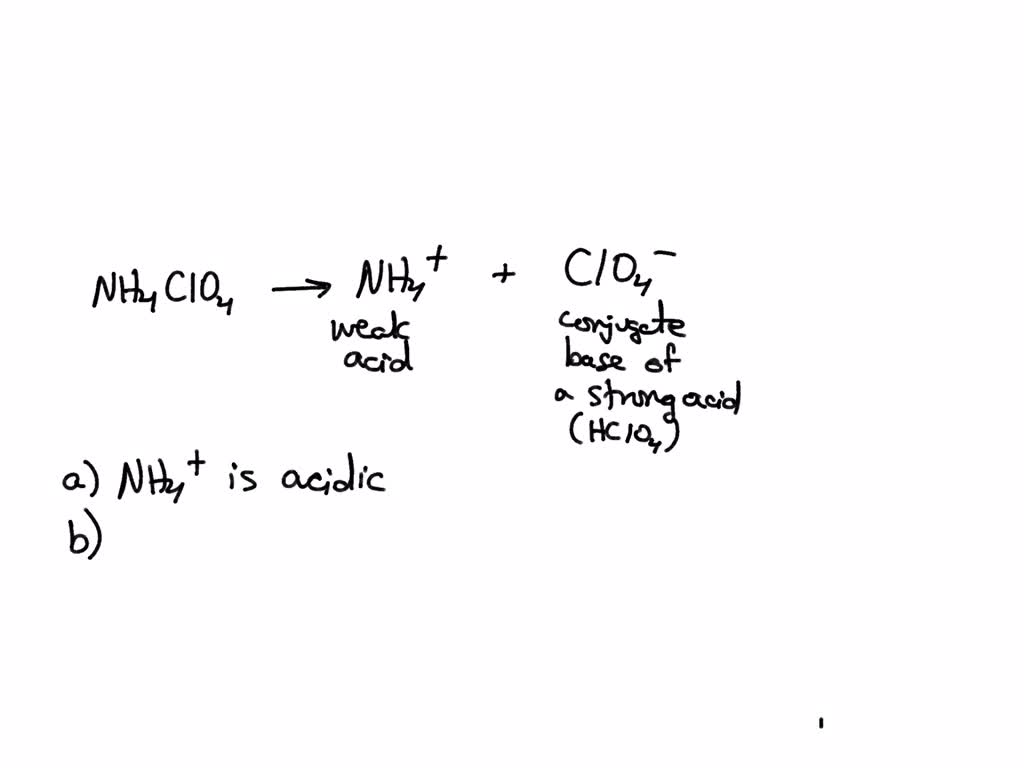
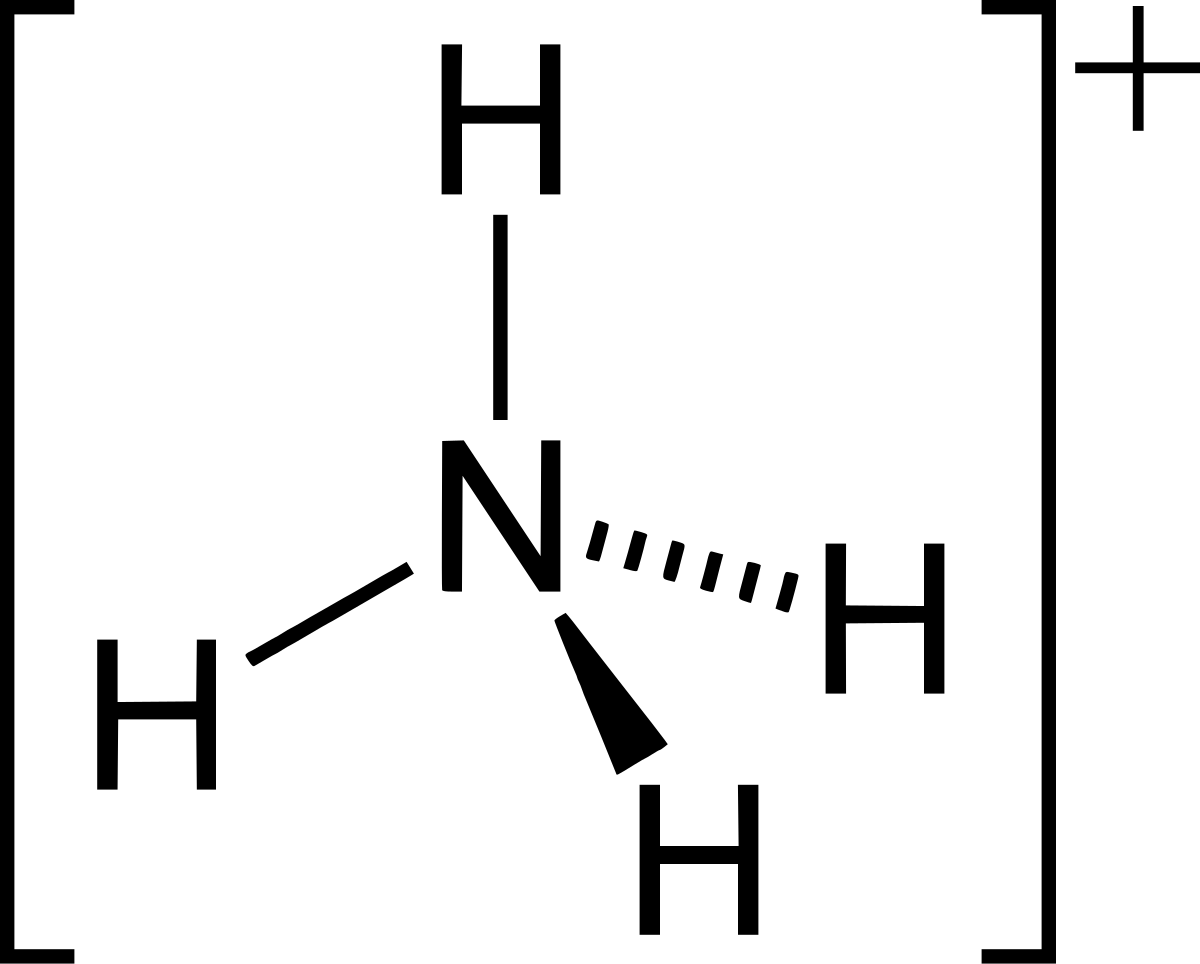
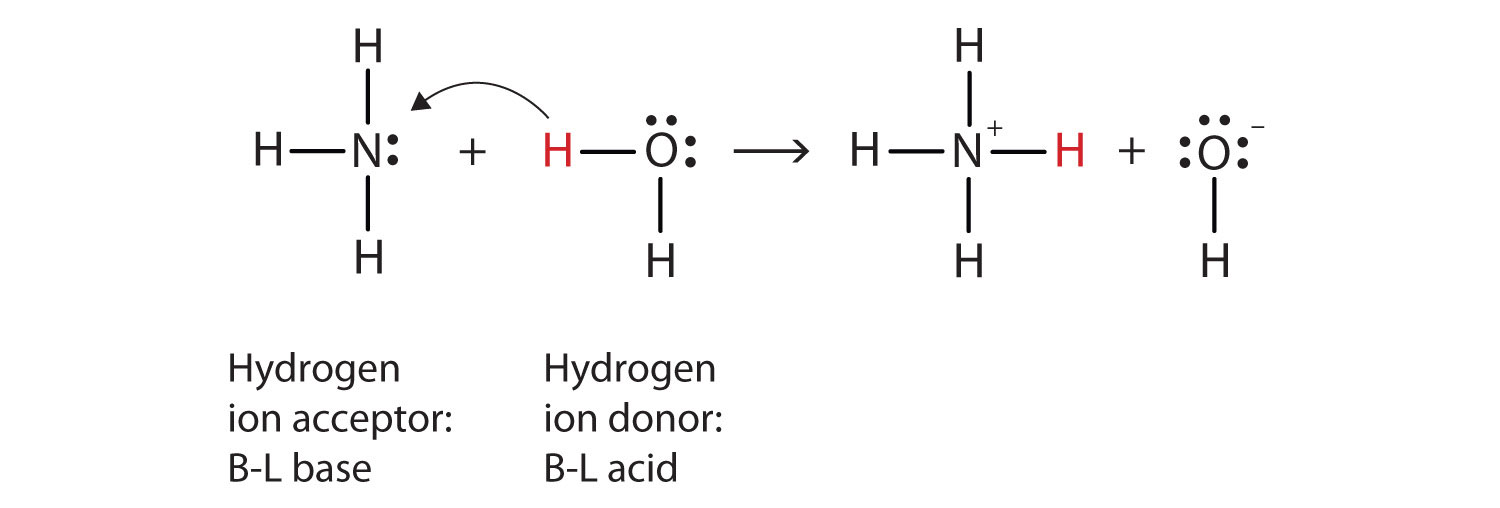
![high school : acid and base] is that ammonium ion a weak acid or base. Because it release OH- when react with water but when I search in Internet, it shows that high school : acid and base] is that ammonium ion a weak acid or base. Because it release OH- when react with water but when I search in Internet, it shows that](https://i.redd.it/kcgf74ac4i151.jpg)
high school : acid and base] is that ammonium ion a weak acid or base. Because it release OH- when react with water but when I search in Internet, it shows that

List of acids, bases and new ammonium-based protic ionic liquids (PILs)... | Download Scientific Diagram

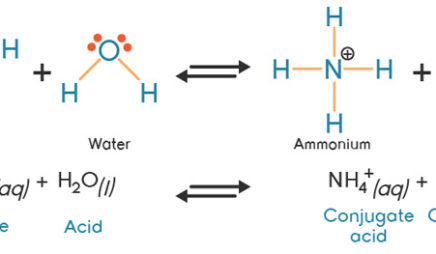
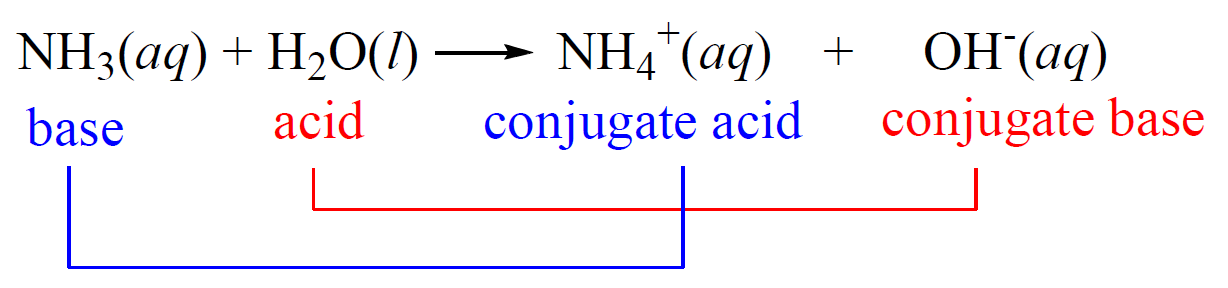
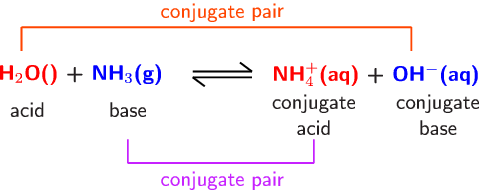
Ammonia is a weak base that reacts with water according to the equation NH(3)(aq)+H(2)O(l)hArrNH(4)^(+)(aq)+OH^(-)(aq) Select the correct option (s) that can increase the moles of ammonium ion in water:

The pH of aqueous solution of ammonium formate is `(pK_(a)` of HCOOH =3.7 and `NH_(3)=4.8)` - YouTube